Cirulli Laboratory
The Cirulli laboratory is committed to understanding mechanisms of cell-cell and cell-matrix interactions in pancreatic islet cell development and function.
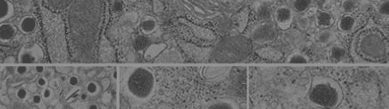
The overall objective of our laboratory is to understand cellular and molecular mechanisms that foster the development and function of pancreatic islets of Langerhans, small clusters of 2,000-3,000 cells scattered throughout the pancreas, that are responsible for the production of such hormones as insulin, glucagon, somatostatin, pancreatic polypeptide, and ghrelin. Special attention in the laboratory is devoted to insulin-producing cells (also known as β-cells) whose function is lost in both type 1 and type 2 diabetes. The specific focus of our research program is to understand the function of highly specialized proteins that regulate cell-cell and cell-matrix interactions in the pancreas. Our previous work has established that these proteins, also referred to as “cell adhesion molecules,” are used by cells not only to aggregate with each other, but also to let cells talk to one another through the exchange of biochemical signals, thereby helping developing cells to decide whether to grow (i.e. increase in numbers) or to differentiate (i.e. mature) into functional adult β-cells.
In an effort to develop strategies of possible translational value to human diabetes, our team follows a multi-pronged approach: on the one hand we are using gene manipulation techniques to uncover the function of select cell adhesion molecules in β-cell development and function, and on the other hand we are using select cell adhesion molecules produced in our lab as biochemical cues to test whether they elicit the maturation of pluripotent stem cells to become insulin-producing cells, and/or to trigger the proliferation and expansion of donor human β-cells, thus increasing their number for cell-based replacement strategies to cure diabetes.
So far, we have identified several cell adhesion molecules that appear to influence cell communication and development in the pancreas. One of such molecules, named Ep-CAM (Epithelial Cell Adhesion Molecule), appears to be used by cells preferentially when they need to replicate (i.e., proliferate) (Cirulli et al. 1998; Vercollone et al., 2015).
Other molecules that the Cirulli lab has defined as important regulators of islet cell development and function include β1 integrin receptors (Cirulli et al., 2000; Diaferia et al, 2013), and members of the chemoattractant proteins Netrins (Yebra et al., 2003; Cirulli & Yebra 2007; Yebra et al., 2011).
More recently, we discovered that aE-catenin, a key component of adhesion complexes, functions as a positive regulator of pancreatic islet cell lineage differentiation by repressing the sonic hedgehog pathway (SHH). We found that deletion of aE-catenin in multipotent pancreatic progenitors results in the loss of adherens junctions, constitutive activation of SHH, decrease in islet cell lineage differentiation, and accumulation of immature Sox9+ progenitors. Pharmacological blockade of SHH signaling in these aE-catenin null Sox9+ pancreatic progenitors rescues this defect, allowing their differentiation into endocrine cells (Jimenez-Caliani et al., 2017). The results uncover crucial functions of aE-catenin in pancreatic islet development and harbor significant implications for the design of β-cell replacement and/or regeneration therapies in diabetes.


Current and Recent Lab Members




Contact Us
UW Diabetes Institute
850 Republican Street, Box 358062
Seattle, WA 98109
Cirulli Office: (206) 685-6638
Laboratory: (206) 616-6090
Vincenzo Cirulli: vcirulli@medicine.washington.edu
Patricia Kensel-Hammes (Lab Manager): pkh21@medicine.washington.edu
Careers: To inquire about Postdoctoral and Graduate Student Openings click on: vcirulli@uw.edu